Customer Support
Product Orders
(866) 246-6990 USA Product orders by FAX: (866) 804-9881 USA Email Customer Service Contact Us Form

D-Stat® Flowable Hemostat
Shown to reduce the frequency of clinically relevant hematoma formation in pulse generator implants1,2,3
Comprised of collagen, thrombin and a buffered diluent, this thick, yet flowable procoagulant facilitates hemostasis by initiating the body’s own clotting mechanisms.
Practical advantages
- Fully sterile kit allows complete preparation within sterile field
- Easy, needle-free mixing
- No pharmacy storage required
The Pocket Protector Study showed a 48% reduction in clinically relevant hematomas1,2,3
The Pocket Protector Study was a prospective, randomized clinical study evaluating the incidence of clinically relevant hematoma formation after pulse generator implants using D-Stat® Flowable Hemostat plus standard of care vs. standard of care alone.4
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
*Statistically significant reduction (p ≤ 0.05) calculated using Fischer’s Exact Test. |
|||
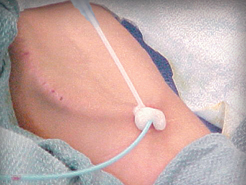
D-Stat® Flowable Hemostat for use in vascular access sites
Applied topically to control bleeding from vascular access sites and percutaneous catheters and tubes.
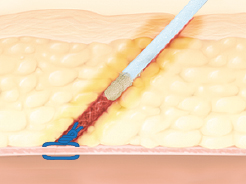
D-Stat® Flowable Hemostat for use in tissue tract hemostasis post vascular closure device
Applied as an adjunct treatment to seal residual oozing of tissue tracts of femoral access sites that have been previously closed by suture/collagen-based hemostatic devices.
References:
- Slotwiner D, Alder S, Fuenzalida C, McCowan R, et al. The Pocket Protector Study: Use of D-Stat® Flowable Hemostat in Pulse Generator Pectoral Pockets Reduces the Rate of Clinically Relevant Hematomas. Circulation. 2007;116:II_678 (the "Pocket Protector Study").
- The Pocket Protector Study evaluated 269 high-risk patients, which were defined as those whose anticoagulation regimens were resuming within 24 hours. These regimens included the administration of one or more of the following medications: Heparin, LMWH, Coumadin®, Plavix®. For patients receiving Coumadin therapy, an INR of < 2.0 was required.
- Shapes of hematomas vary depending on the tissue planes that are dissected in. In the Pocket Protectory Study the following formula was used to identify clinically relevant pocket hematomas. "Length, width, and height of any palpated hematoma to approximate the amount of blood present using the following formula for the volume of an ellipsoid: V = (1/6) π (L)(W)(H). Where: V = Volume, L = Length (cephalead/caudal), W = Width (medial/lateral), and H = Height (anterior to pulse generator)." "Based on this formula, the volume of blood present in a hematoma measuring 8 x 1 x 2 (cm) (L,W,H) is approximately 8.5ml, therefore this volume shall serve as the minimum volume for a clinically relevant hematoma."
- The Pocket Protector Study defined standard of care as compression, electrocautery and/or untreated cotton pledgets.
The D-Stat flowable is indicated for use as an adjunct treatment in sealing residual oozing of tissue tracts of femoral access sites that have been previously closed by suture/collagen-based hemostatic devices.
D-Stat flowable is indicated for use in high-risk, anti-coagulated patients undergoing implantation of a pulse generator (e.g., pacemaker or ICD) to reduce the frequency of clinically relevant hematoma formation in the prepectoral pocket. High-risk patients are defined as those whose anticoagulation regimens will resume within 24 hours of implant. Clinically relevant hematomas are defined as those that result in an alteration in the standard of care resultant of hematoma formation including alteration (i.e. suspension or discontinuation) of the anticoagulant therapy regimen (Heparin, LMWH, Coumadin or Plavix), application of a compression bandage and evacuation of the hematoma.
CONTRAINDICATIONS: The D-Stat is contraindicated in persons with known sensitivity to bovine-derived materials.
WARNINGS: The effectiveness of D-Stat flowable to reduce the frequency of prepectoral pocket hematomas in patients on Coumadin therapy that have an INR >2.0 has not been established.
Do not inject the D-Stat into blood vessels. Extensive intravascular clotting and even death may result. Intravascular delivery of flowable hemostatic agents has been reported. Appropriate imaging should be used when delivering hemostatic agents near vasculature.
Do not use the D-Stat in the closure of skin incisions because it may interfere with the healing of the skin edges due to mechanical interposition of collagen.
Do not use the D-Stat if pulsatile arterial blood flow is observed at the femoral artery access site previously closed by suture/collagen-based hemostatic device. This may indicate incomplete closure of the arteriotomy and may result in inadvertent intraarterial injection of the D-Stat. The acute onset of severely diminished or absent pulses in the limb treated with the D-Stat may indicate that inadvertent intraarterial injection has occurred. If this is suspected, immediately perform appropriate diagnostic and therapeutic procedures for thrombus dissolution/removal.
Do not use the D-Stat to treat residual oozing of a femoral artery access site tissue tract if a venous sheath was placed directly adjacent to the site. Inadvertent venous injection may occur.
The safety and effectiveness of D-Stat has not been established for use to control bleeding following organ or tissue biopsies. Use of D-Stat in this situation has been reported to result in pain, seizures, bile retention, tissue necrosis, vascular occlusion and death.
WARNING:
SEVERE
BLEEDING AND THROMBOSIS COMPLICATIONS
- THROMBIN-JMI® can cause fatal severe bleeding or thrombosis. Thrombosis may
result from the development of antibodies against bovine thrombin. Bleeding may result from the
development of antibodies against factor V. These may cross-react with human factor V and lead
to
its deficiency.
- Do not re-expose patients to THROMBIN-JMI® if there are known or suspected
antibodies to bovine thrombin and/or factor V.
- Monitor patients for abnormal coagulation laboratory values, bleeding, or thrombosis.
PRECAUTIONS: D-Stat is not intended to be used as a primary means of hemostasis for the femoral artery access site. The D-Stat should not be used in the presence of infection and it should be used with caution in contaminated areas of the body as it may potentiate bacterial growth.
ADVERSE EFFECTS: Potential adverse effects (in alphabetical order) that may be associated with the D-Stat Flowable hemostat include, but are not limited to the following: Adhesion formation, Allergic reaction, Bleeding, Death, Embolization, Fluid Encapsulation, Foreign Body Reaction, Hematoma, Hypotension, Immunological Reaction, Improper Healing, Infection, Mass Formation, Nerve Injury, Pain, Paralysis, Skin Discoloration, Tissue Injury, Vasovagal Reaction, Wound Dehiscence.
Refer to the Instructions for Use for a complete listing of the indications, contraindications, warnings and precautions. Information in this material is not a substitute for the product Instructions for Use.
CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician.
Not all products are available in all regions. Please contact customer service to confirm availability in your region.
Plavix is a registered trademark of Sanofi-Societe Anonyme.
Coumadin is a registered trademark of Bristol-Myers Squibb Pharma Company.
THROMBIN-JMI is a registered trademark of King Pharmaceuticals Research and Development, LLC.
Teleflex, the Teleflex logo and D-Stat are trademarks or registered trademarks of Teleflex Incorporated or its affiliates, in the U.S. and/or other countries. MC-004466 Rev 0.1
Revised: 02/2020.