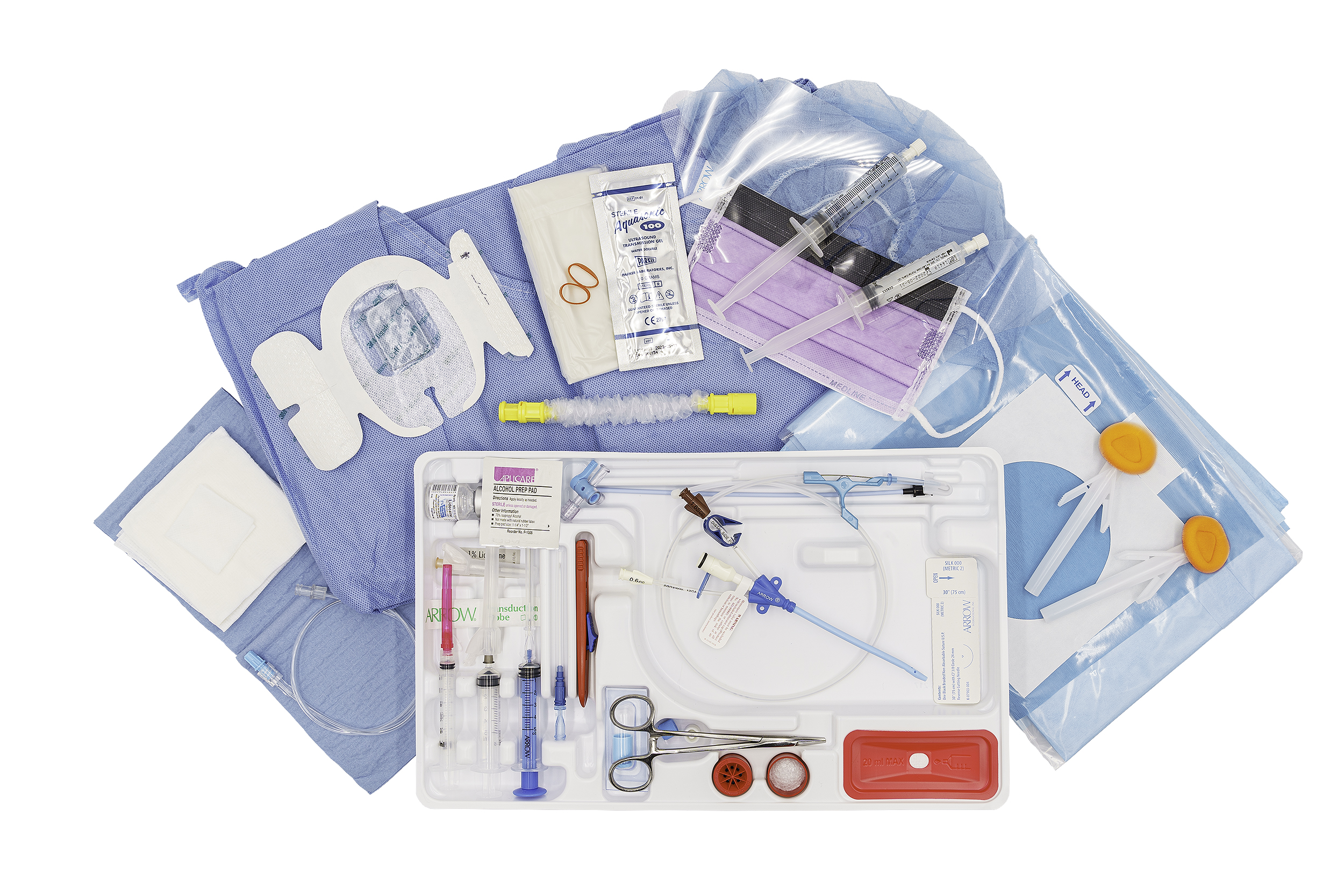

Arrow GlideWheel™ Advancer
Provides tactile feedback + finer control

Nitinol wire
Is kink-resistant compared to stainless steel

Transducer cover
To support guidelines for ultrasound insertion

Pre-filled sterile saline syringe
One pre-filled sterile saline syringe per lumen now included inside the kit

Extra ChloraPrep® Skin Prep
Extra applicator now included in each kit