
Arrow® ErgoPack® Complete System
You needCompleteconfidencein your protected central line access.
What if you could improve efficiency and reduce infections across multiple types of vascular access procedures simply by changing to a kit that provides maximal barrier protection?1
You need the right solution for optimal catheter insertion efficiency. We’ve updated our maximal barrier Central Venous Catheter (CVC), Multi-Access Catheter (MAC), Percutaneous Sheath Introducer (PSI), Hemodialysis and Large Bore insertion systems based on clinician feedback. It puts all the necessary components in an intuitive configuration to help optimize your workflow, while also complying with CDC, SHEA, INS, and OSHA guidelines.2-5 And most importantly, it continues to fight against central line-associated bloodstream infections (CLABSI), protecting your patients and improving procedures.
See the Arrow® ErgoPack® Complete Kit in action
Explore a virtual tour of the Arrow® ErgoPack® Complete Kit and learn how it can help you increase efficiency and patient safety.
Check out what’s different

Nitinol wire
Kink-resistant compared to stainless steel
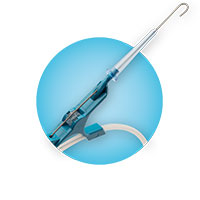
Arrow® GlideWheel™ Advancer
Provides tactile feedback + finer control

Transducer cover
Supports guidelines for ultrasound insertion
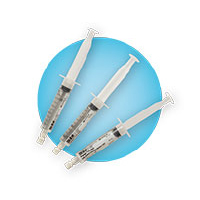
Pre-filled sterile saline syringe
One per lumen now included inside the kit

Extra ChloraPrep®
skin prep
Extra applicator in each kit

DuPont™ Tyvek® sterile packaging
Resists tearing and punctures to protect sterility
Everything for a reason
We’re always thinking about what matters to you and your patients — it’s our entire product development philosophy, methodology and culture.




Take the next step
Download our free educational and product material:
References:
- Raad, II, Hohn DC, Gilbreath BJ, et al. Prevention of central venous catheter-related infections by using maximal sterile barrier precautions during insertion. Infection Control and Hospital Epidemiology. 1994;15(4 Pt 1): 231-238.
- O’Grady NP, Alexander M, Burns LA, et al. Guidelines for the Prevention of Intravascular Catheter-Related Infections, 2011 (Revised 2017). 2. Atlanta, GA: Centers for Disease Control and Prevention; 2017.
- Buetti, N., Marschall, J., Drees, M., Fakih, M., Hadaway, L., Maragakis, L., Mermel, L. Strategies to prevent central line associated bloodstream infections in acute-care hospitals: 2022 Update. Infection Control & Hospital Epidemiology. 2022; 43(5): 1-17. doi:10.1017/ice.2022.87.
- Gorski, Lisa A., et al. Infusion therapy standards of practice. Journal of Infusion Nursing. 44.1S (2021): S1-S224.
- Occupational Safety & Health Administration Regulations (Standards – 29 CFR). Part 1910.1030: Bloodborne pathogens. Occupational Safety & Health Administration Web site. https://www.osha.gov/laws-regs/regulations/standardnumber/ 1910/1910.1030. Accessed on February 12, 2020.
- Rupp ME, Lisco SJ, Lipsett PA, et al. Effect of a Second-Generation Venous Catheter Impregnated with Chlorhexidine and Silver Sulfadiazine of Central Catheter-Related Infections. Annals of Internal Medicine. 2005; 143: 570-80. Sponsored by Arrow (Teleflex).
- Lorente L, Lecuona M, Jimenez A, et al. Chlorhexidine-silver sulfadiazine-impregnated venous catheters save costs. American Journal of Infection Control. 2014; 42: 321-4.
- Lorente L, Lecuona M, Jimenez A, et al. Cost/benefit analysis of chlorhexidine-silver sulfadiazine-impregnated venous catheters for femoral access. American Journal of Infection Control. 2014; 42: 1130-2.
- Lorente L, et al. Chlorhexidine-silver sulfadiazine-impregnated venous catheters are efficient even at subclavian sites without tracheostomy. American Journal of Infection Control. 2016; 44(12): 1526-29.
Contraindication:
The Arrowg+ard Blue®and Arrowg+ard Blue Plus®Catheters are contraindicated for patients with known hypersensitivity to chlorhexidine and silver sulfadiazine and/or sulfa drugs.
Rx Only. CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. See Instructions for Use for detailed information regarding the instructions for use, contraindications, potential adverse events, warnings and cautions.
DuPont™, the DuPont Oval Logo, and all trademarks and service marks denoted with ™, ℠ or ® are owned by affiliates of DuPont de Nemours, Inc. unless otherwise noted.
ChloraPrep is a trademark or registered trademark of Becton, Dickinson and Company.
Information in this material is not a substitute for the product Instructions for Use. Not all products may be available in all countries. Please contact your local representative. Revised: 6/2023.
Teleflex, the Teleflex logo, Arrow, Arrowg+ard Blue, Arrowg+ard Blue Plus, ErgoPack, and GlideWheel are trademarks or registered trademarks of Teleflex Incorporated or its affiliates, in the U.S. and/or other countries. All other marks are trademarks or registered trademarks of their respective owners.
© 2023 Teleflex Incorporated. All rights reserved. MC-006498 Rev 1.1

