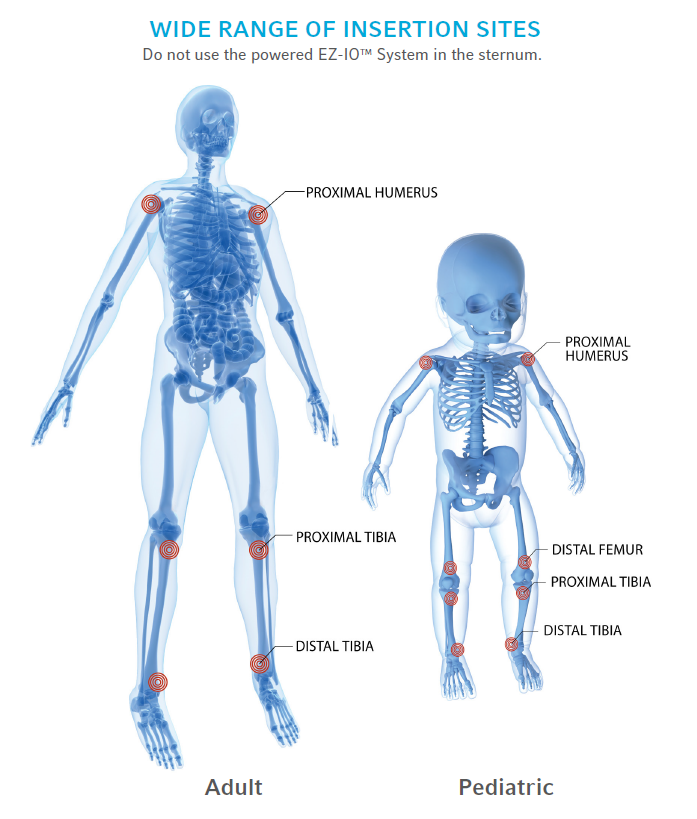
References:
- Teleflex Internal Data on File 2024.
- Davidoff J, Fowler R, Gordon D, Klein G, Kovar J, Lozano M, Potkya J, Racht E, Saussy J,
Swanson E, Yamada R, Miller L. Clinical evaluation of a novel intraosseous device for adults:
prospective, 250-patient, multi-center trial. JEMS 2005;30(10):s20-3. Research sponsored by
Teleflex Incorporated.
- Montez D, Puga T, Miller LJ, et al. Intraosseous Infusions from the Proximal Humerus Reach the
Heart in Less Than 3 Seconds in Human Volunteers. Annals of Emergency Medicine 2015;66(4S):S47.
Research sponsored by Teleflex Incorporated.
- Cooper BR, Mahoney PF, Hodgetts TJ, Mellor A. Intra-osseous access (EZ-IO™) for
resuscitation: UK military combat experience. J R Army Med Corps. 2007;153(4):314-316
- Hoskins SL, Nascimento P Jr., Lima RM, Espana-Tenorio, JM, Kramer GC. Pharmacokinetics of
intraosseous and central venous drug delivery during cardiopulmonary resuscitation.
Resuscitation 2011; doi:10.1016/j.resuscitation.2011.07.041. Research sponsored by Teleflex
Incorporated. (preclinical study)
- Hoskins SL, Zachariah BS, Copper N, Kramer GC. Comparison of intraosseous proximal humerus and
sternal routes for drug delivery during CPR. Circulation 2007; 116:II_993. Research sponsored by
Teleflex Incorporated. (preclinical study)
*The Arrow™ EZ-IO™ Intraosseous Vascular Access System is indicated for intraosseous access anytime in which vascular
access is difficult to obtain in emergent, urgent or medically necessary cases for up to 24 hours. For patients ≥ 12
years old, the device may be extended for up to 48 hours when alternate intravenous access is not available or
reliably established. Refer to the Instructions for Use for a complete listing of indications, contraindications,
warnings, precautions, and approved insertion sites.
**Compared to single lumen Central Venous Catheters
(CVCs).
†Based on Adult Proximal HumerusBased on Adult Proximal Humerus
EZ-IO™ insertion data.
Rx Only.
CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician.
The Arrow™ EZ-IO™ Needle Sets are single use only. Failure to comply with this
warning may result in
serious medical
consequences (e.g., life-threatening infection) and reduced performance (e.g., blunted needles). Please refer to the
Instructions for Use for complete warnings, indications, contraindications, precautions, and potential
complications.
Needle Vise is a trademark or registered trademark of Atrion Medical Products,
Inc.
Teleflex, the Teleflex logo, Arrow, EZ-Connect, EZ-IO and EZ-Stabilizer are trademarks or
registered
trademarks of Teleflex Incorporated or its affiliates, in the U.S. and/or other countries. MC-000220Rev3