Customer Support
South Africa Office Address
Teleflex Medical (Pty) Ltd. 1st Floor, Block C, Stoneridge Office Park, 8 Greenstone Place, Greenstone, Edenvale, 1609, Johannesburg, South Africa Phone: +27(0)11 807 4887 Fax: +27 (0)11 807 4994
South Africa Mail Address
Teleflex Medical (Pty) Ltd. P.O. Box 6882, Greenstone, 1616
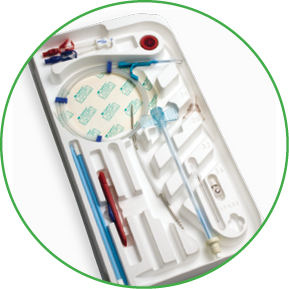
Email Teleflex Medical Ltd.Arrow Edge Antegrade-Tunneled Chronic Hemodialysis Catheter
Staggered V-Tip Design for Optimal Performance
The Arrow Edge Catheter is designed to provide optimal performance by delivering high flow rates, up to 400 cc per minute, at low pressures.1 The crescent-shape of the catheter 's distal V- tip is designed to promote blood flow even if the tip is resting against the vessel wall.
- Innovative V-tip design and catheter tip orientation minimise the risk of recirculation when placed in the right atrium2
- Compatible with broad spectrum of antiseptic agents, including alcohol and iodine3
- Kits feature Arrow SmartSeal Hemostatic Peelable Dialysis Sheath
References:
- In vitro test performed by an independent laboratory (Citech, Plymouth Meeting, PA). Laboratory test results may not be indicative of clinical performance.
- Caridi J, Ross E, Aspilcueta A, Wiley S. The cannon-catheter-a prospective analysis. J of Vasc and Interv Radiol. 2010;21:1588-1590. The Arrow Edge® Catheter is the antegrade version of and has the same tip design as the retrograde Arrow® Cannon® Catheter. Both catheters are designed for placement in the mid-right atrium.
- Alcohol, alcohol-based solutions (e.g., Hibiclens®, ChloraPrep®), iodine-based solutions (Povidone-Iodine), PEG-based ointments (e.g., Bactroban®), hydrogen peroxide or ExSept Plus® are accepted for use with this catheter.