Customer Support
IDA Business and Technology Park
Dublin Road, Athlone
Co Westmeath
Ireland
Phone: +353 (0) 9 06 46 08 00
Fax: +353 (0) 14 37 07 73
Email Europe, Middle East, Africa Regional
Office
United Kingdom
Teleflex
Oakfield House
59 Hill Avenue
Amersham
HP6 5BX
Phone: +44 (0)1494 53 27 61
Fax: +44 (0)1494 52 46 50
Email Teleflex Medical
South Africa Office Address
Teleflex Medical (Pty) Ltd.
1st Floor, Block C,
Stoneridge Office Park,
8 Greenstone Place,
Greenstone, Edenvale, 1609,
Johannesburg, South Africa
Phone: +27(0)11 807 4887
Fax: +27 (0)11 807 4994
South Africa Mail Address
Teleflex Medical (Pty) Ltd.
P.O. Box 6882, Greenstone, 1616
Email Teleflex Medical Ltd.
Arrowg+ard Blue Plus CVC
Help protect your patients from catheter-related complications.
The Arrowg+ard Blue Plus CVC is the only full-spectrum antimicrobial CVC that protects against:
- Gram-positive bacteria
- Gram-negative bacteria
- Fungi
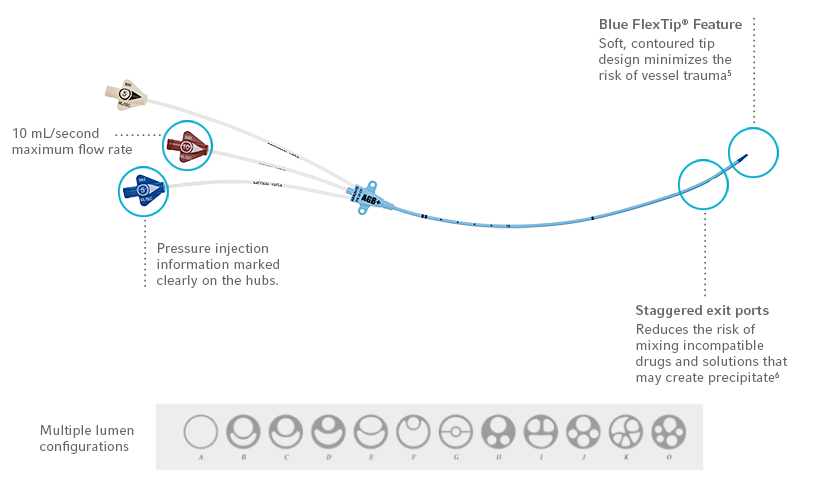
Arrowg+ard Blue Plus Protection is chemically bonded to the catheter, providing antimicrobial protection on the catheter surface.
- Complete coverage – extension lines, hub and catheter protected1
- In vitro performance – proven effectiveness against common antibiotic resistant “super bugs”2
Meets Guidelines and Recommendations:
- Centers for Disease Control (CDC)*3
- The Society for Healthcare Epidemiology of America (SHEA)**4
- INS5
- ESPEN, Europe6
- Epic3, United Kingdom7
- RCN, United Kingdom8
- KRINKO, Germany9

References:
- https://www.accessdata.fda.gov/cdrh_docs/pdf/K993691.pdf
- Gupta N, Weber H, Moss S, Gaire-Patel K. Are antibiotic resistant “super bugs” a real challenge to antimicrobial central venous catheter performance? AVA 2014.
- O’Grady NP, Alexander M, Burns LA, Dellinger P, Garland J, Heard SO et al Guidelines for the Prevention of Intravascular Catheter-related Infections, 2011. The Centers for Disease Control. www.cdc.gov/hicpac/pdf/guidelines/bsi-guidelines-2011.pdf
- SHEA Compendium https://www.shea-online.org/index.php/practice-resources/priority-topics/compendium-of-strategies-to-prevent-hais
- Gorski LA, Hadaway L, Hagle M, Broadhurst D, Clare S. Infusion therapy standards of practice. Journal of Infusion Nursing. 8th Edition Revised 2021
- Pittiruti M, Hamilton H, BiffiR, MacFieJ, PertkiewiczM, Espen. ESPEN Guidelines on Parenteral Nutrition: central venous catheters (access, care, diagnosis and therapy of complications). Clinical nutrition. 2009;28(4):365-377
- Loveday HP, Wilson JA, Pratt RJ, et al. epic3: national evidence-based guidelines for preventing healthcare-associated infections in NHS hospitals in England. J Hosp Infect. 2014;86 Suppl 1:S1-70.
- Standards for infusion therapy. 4th ed. London: Royal College of Nursing; 2016.
- Kommission für Krankenhaushygiene und Infektionsprävention (RKI KRINKO). Prävention von Infektionen, die von Gefäßkathetern ausgehen. Bundesgesundheitsbl. 2016;60(2):171-206.doi:10.1007/s00103-016-2487-4
- Rosenbauer KA, Herzer JA. Surface morphology and tensile force at breaking point or different kinds of intravenous catheters before and after usage. Scan Electron Microsc. 1981;(Pt 3):125-30.
- Collins JL, Lutz RJ. In vitro Study of Simultaneous Infusion of Incompatible Drugs in Multilumen Catheters. Heart & Lung. 1991; 20(3):271-7.
**Use such catheters in the following instances: i. Hospital units or patient populations have a CLABSI rate above institutional goals despite compliance with basic CLABSI prevention practices. Some evidence suggests that use of antimicrobial CVCs may have no additional benefit in patient care units that have already established a low incidence of catheter infections. ii. Patients have limited venous access and a history of recurrent CLABSI. iii. Patients are at heightened risk of severe sequelae from a CLABSI (e.g, patients with recently implanted intravascular devices, such as a prosthetic heart valve or aortic graft).
Contraindication:
The Arrowg+ard Blue Plus CVC is contraindicated for patients with known hypersensitivity to chlorhexidine and silver sulfadiazine and/or sulfa drugs.
Teleflex, the Teleflex logo, Arrow, Arrowg+ard Blue Plus, and Blue FlexTip are trademarks or registered trademarks of Teleflex Incorporated or its affiliates, in the U.S. and/or other countries. All other trademarks are trademarks of their respective owners. MCI-100375