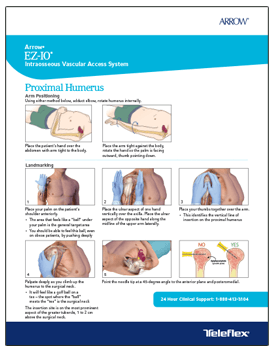
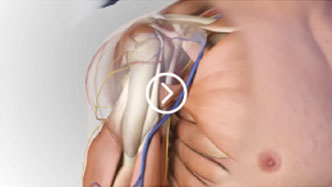
Proven
More than 90 published case studies or clinical trials,8 learn more
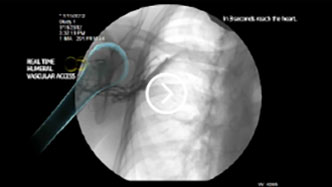
Fast
Achieves vascular access within 10 seconds2
Effective
97% first-attempt access success rate3
It's time to think of intraosseous access earlier.
Procedure Time
The median procedure time for PIV access in DVA patients is 8.8 minutes. However, the average Arrow EZ-IO System procedure time is 5.0 minutes.10¥